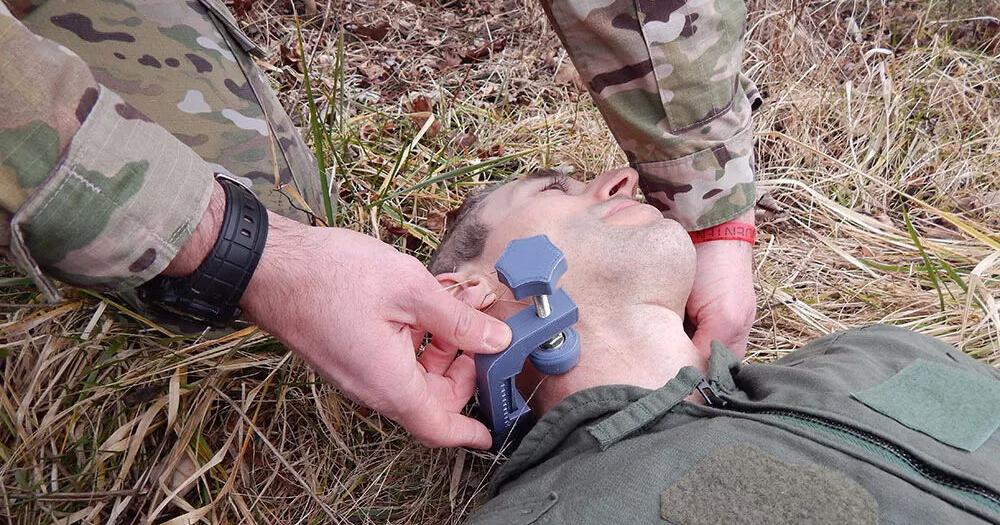
Researchers at Cedarville University in Cedarville, Ohio, have created a medical device aimed at slowing deadly bleeding from traumatic neck wounds, a problem that can turn fatal in minutes, and the U.S. Food and Drug Administration recently engaged with the issue. The work out of Cedarville focuses on a practical tool first responders and surgeons could use when seconds count. This article traces what the device means in plain terms, why neck bleeding is uniquely dangerous, and how regulatory attention from the FDA matters for getting tools into real-world use.
Neck trauma is deceptively simple to describe and brutally hard to treat. A few centimeters of bleeding in the wrong place can compress airways, damage major vessels, and turn a survivable injury into a fatal one within minutes. That window of time is where innovations matter most: devices that stall hemorrhage and give medics a chance to secure an airway or transport a patient to surgery change outcomes.
Cedarville University researchers focused on those exact minutes. Rather than chasing a flashy cure, they built a device meant to be fast to deploy and effective at cutting blood loss from deep or awkward neck wounds. The aim is straightforward: reduce bleeding just enough to prevent immediate death while more definitive care is arranged.
Practicality drives good emergency gear. From what Cedarville is reporting, the device prioritizes speed, ease of use, and compatibility with existing emergency procedures. First responders often work in cramped, chaotic scenes; anything that requires precise setup or delicate handling can be impractical. So a device that can be applied quickly and reliably stands a better chance of actually saving lives in the field.
There are engineering and medical hurdles to clear when designing around the neck. The area packs arteries, veins, nerves, and the windpipe into a tight space, so any tool must stop bleeding without creating new dangers. That balance—control hemorrhage while avoiding compression of the airway or major nerves—is the core design challenge the Cedarville team addressed.
The U.S. Food and Drug Administration recently drew attention to technologies that control catastrophic bleeding, which matters because federal review shapes whether tools move from prototype to ambulance cabinets. FDA involvement signals that regulators are aware of the clinical need and the risks involved, and it creates a pathway for testing safety and effectiveness under controlled conditions. For inventors, that pathway is essential; for clinicians, it’s the assurance they can rely on the device; for patients, it’s the best route to better outcomes.
Bringing a new hemorrhage-control device into practice requires more than good engineering and regulatory clearance. Training, supply chain, cost, and integration into protocols all determine whether an innovation actually gets used. Cedarville’s next steps likely include real-world testing with emergency teams, refinement based on feedback, and scaled manufacturing plans that keep the price point reasonable for widespread adoption.
At the same time, hospitals and EMS services watching the space want clear evidence that tools improve survival without introducing extra risk. That means clinical data showing not just that the device stops bleeding but that it does so while protecting the airway and surrounding structures. Manufacturers and researchers who deliver transparent, reproducible results are the ones that earn a place in treatment guidelines.
Innovations like this matter because they attack a specific, time-critical problem. If the Cedarville device proves reliable in practice and clears regulatory hurdles, it could become a simple addition to emergency kits that changes how crews handle neck trauma. That kind of change won’t be headline-grabbing, but it could tip survival odds in favor of patients when every second counts.